 
L-glutamate in a transamination reaction catalyzed byĪspartate aminotransferase 2, and is transformed toĢ-oxoglutarate are transported across the inner mitochondrial membrane. Oxaloacetate cannot cross the mitochondrial membrane. Malate dehydrogenase 2, NAD + is reduced to NADH and a proton is released Malate is then converted back to oxaloacetate by Mitochondrial 2-oxoglutarate/malate transporter simultaneously imports malate into the mitochondrial matrix from the cytosol and exportsĢ-oxoglutarate from the matrix to the cytosol. Mitochondrial 2-oxoglutarate/malate transporter.Ĭytosolic malate dehydrogenase catalyzes the transfer of two electrons from Mitochondrial aspartate glutamate transporter 1 and The shuttle consists of six key component proteins, There are two well established shuttles for the transport of reducing equivalents described for vertebrates, the The shuttle involves a reaction between NADH and an oxidized substrate in the cytosol, the reduced substrate is transported into the mitochondrion and subsequently reoxidized by the electron transfer chain. 
Malate-aspartate shuttle is a biochemical entity that facilitates the translocation of electrons across the membrane. 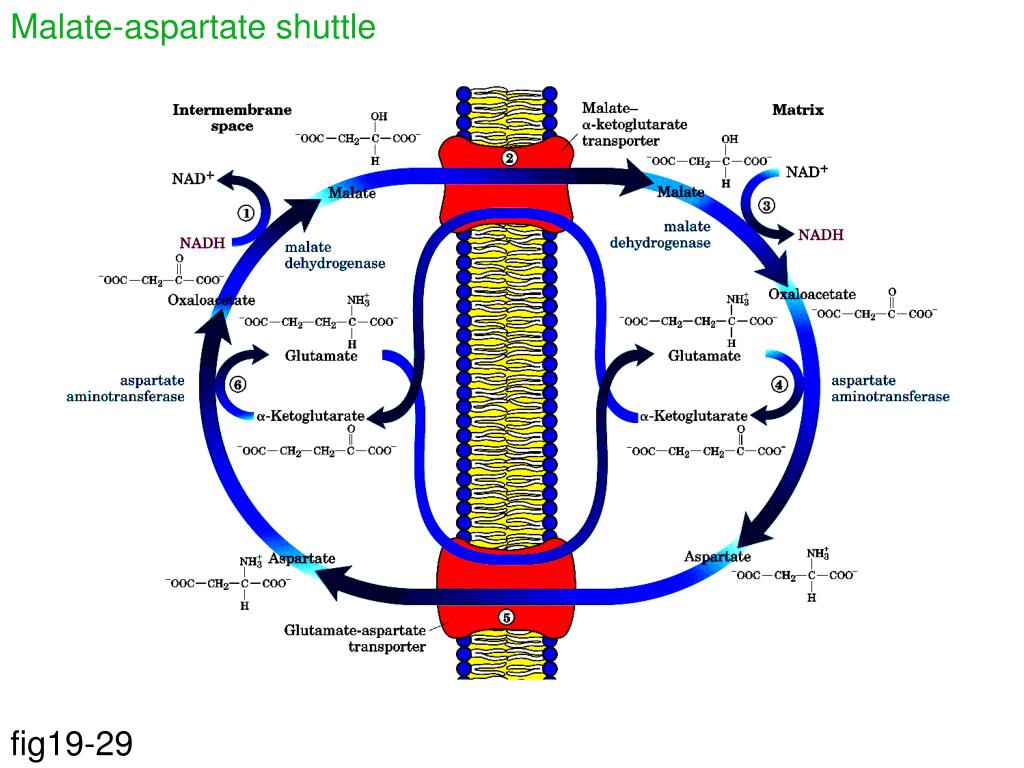
NADH cannot traverse the inner mitochondrial membrane and cytosolic reducing equivalents (hydrogen atoms) must be transferred to the mitochondria across the membrane via metabolite shuttles that operate between the two compartments NADH is the reducing equivalent that supplies electrons to the electron transport chain to generate ATP. NAD + and the NADH oxidation process in the cell occurs primarily via mitochondrial respiration. NADH must be continuously balanced within the cell to maintain the pathways of anabolism and catabolism. Generation of Precursor Metabolites and Energy → Malate/L-aspartate shuttle pathway BioCyc ID: MALATE-ASPARTATE-SHUTTLE-PWY The capacity of the various tumor lines for the reoxidation of cytoplasmic NADH via the malate-aspartate shuttle approaches 20% of the total respiratory rate of the cells and thus appears to be sufficient to account for the mitochondrial reoxidation of that fraction of glycolytic NADH not reoxidized by pyruvate and lactate dehydrognenase in the cytoplasm.If an enzyme name is shown in bold, there is experimental evidence for this enzymatic activity. The maximal rate of the malate-aspartate shuttle was established by addtion of arsenite to inhibit the mitochondrial oxidation of the pyruvate formed from added lactate. Compounds that inhibit the carrier-mediated entry of malate into mitochondria, such as butylmalonate, benzenetricarboxylate, and iodobenzylmalonate, also inhibited the accumulation of pyruvate from added L-lactate by the tumor cells. The involvement of the respiratory chain in the reoxidation of cytoplasmic NADH was demonstrated by the action of cyanide, rotenone, and antimycin A, which strongly inhibited the formation of pyruvate from added L-lactate. Reoxidation of cytoplasmic NADH thus generated was completely inhibited by the transaminase inhibitor aminooxyacetate. All the tumor cells examined showed mitochondrial reoxidation of cytoplasmic NADH, as evidenced by the accumulation of pyruvate when the cells were incubated aerobically with L-lactate. The activity of the malate-aspartate shuttle for the reoxidation of cytoplasmic reduced nicotinamide adenine dinucleotide (NADH) by mitochondria was assessed in six lines of rodent ascites tumor cells (two strains of Ehrlich ascites carcinoma, Krebs II carcinoma, Novikoff hepatoma, AS-30D hepatoma, and L1210 mouse leukemia). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
